N95 vs KN95 Masks: What Healthcare Professionals Need to Know
With various respirator options available to Canadian healthcare workers, clinic administrators, and frontline staff, selecting the right protective equipment can be challenging. Many purchasing managers ask: Are N95 and KN95 masks interchangeable? What distinguishes these two respirator types?
At Surgo Surgical Supply, we're committed to helping you make informed decisions about respiratory protection. This comprehensive guide breaks down the key differences, similarities, and selection criteria for N95 and KN95 respirators.

Understanding the Standards
The primary distinction between N95 and KN95 respirators lies in their certification standards and the regulatory bodies that approve them. While both are designed to filter at least 95% of airborne particles, they operate under different regulatory frameworks.
Certification Authorities
N95 Masks: Meet the American NIOSH (National Institute for Occupational Safety and Health) standard under 42 CFR Part 84. These respirators are certified for use in the United States and widely recognized as the gold standard for respiratory protection.
KN95 Masks: Comply with Chinese GB2626-2006 standards administered by Chinese regulatory authorities. According to the CDC, KN95 respirators are among the most widely available options that meet international filtration standards.
Why Standards Matter
The certification standard determines the testing protocols, quality control requirements, and regulatory oversight that each respirator must meet. While both N95 and KN95 designations indicate 95% filtration efficiency, the testing methods and additional requirements differ between regulatory systems.
For Canadian healthcare facilities, understanding these distinctions helps procurement managers make informed decisions about respiratory protection programs, particularly when supply chain considerations come into play.
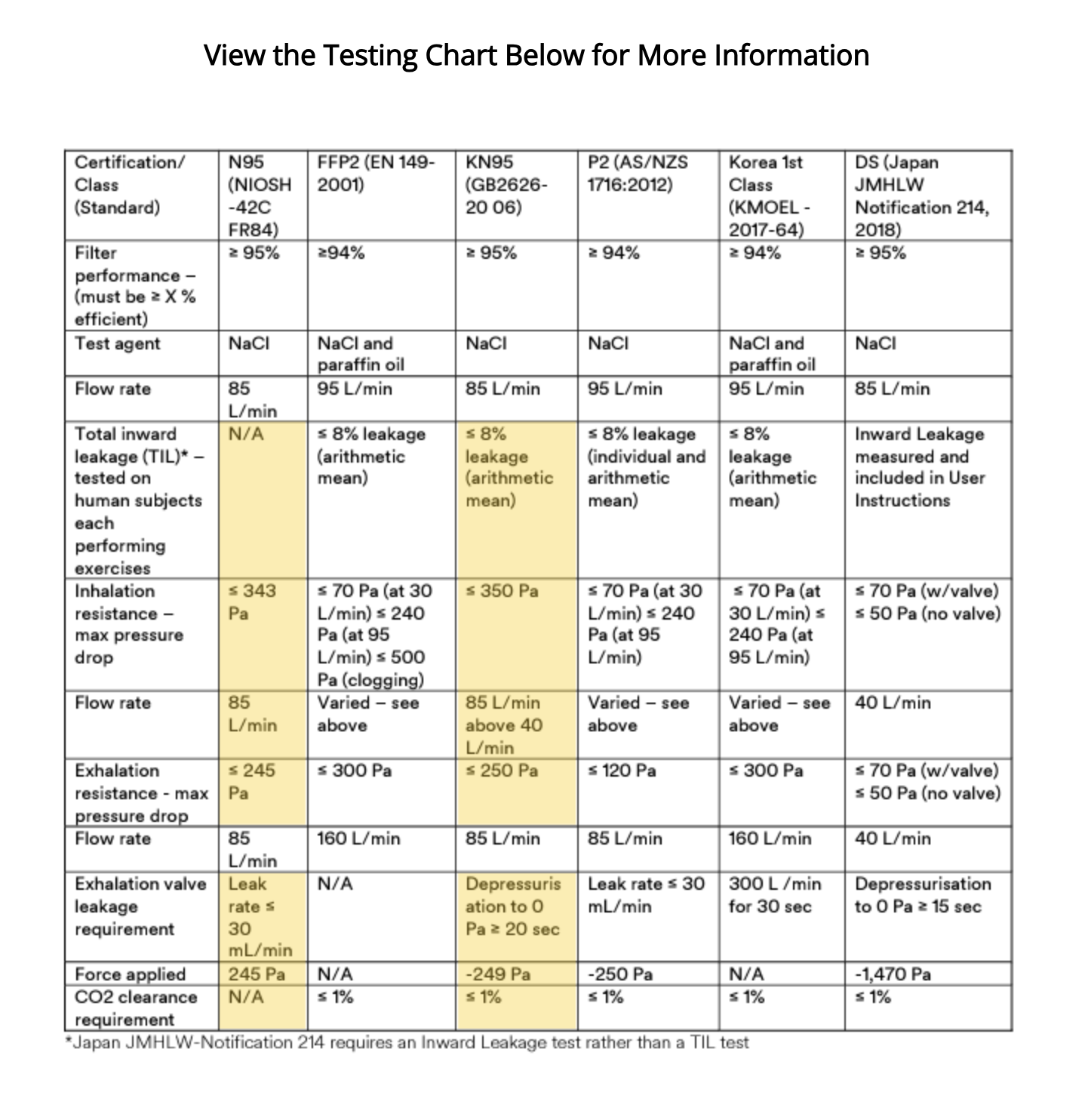
Comparing Performance Requirements
Both N95 and KN95 respirators are designed to filter at least 95% of airborne particles as small as 0.3 microns. However, there are some notable differences in testing protocols and performance requirements.
| Requirement | N95 (NIOSH) | KN95 (GB2626) |
|---|---|---|
| Filtration Efficiency | ≥95% at 0.3 microns | ≥95% at 0.3 microns |
| Fit Testing During Certification | Not required during approval process | Mandated on human subjects |
| Inhalation Resistance | ≤343 Pa (more stringent) | ≤350 Pa |
| Exhalation Resistance | ≤245 Pa (more stringent) | ≤250 Pa |
| Certification Body | NIOSH (U.S.) | Chinese regulatory authority |
| Regulatory Recognition | U.S. federal standard | Chinese national standard |
Fit Testing Requirements
KN95 Standard: The KN95 standard mandates fit testing on human subjects during the certification process. This means manufacturers must demonstrate that their KN95 respirators can achieve proper seal on actual test subjects as part of the approval process.
N95 Standard: The N95 standard does not require fit testing on humans during the approval process. However, OSHA regulations still mandate workplace fit testing for employees who wear N95 respirators as part of their job duties.
Practical Implication: Regardless of which respirator type you choose, implementing fit testing protocols for healthcare workers remains essential for ensuring proper protection.
Breathability Differences
N95 certification includes slightly more stringent requirements for airflow resistance, which translates to marginally easier breathing through the mask during extended wear. The difference is measured in pascals (Pa):
- N95 Inhalation Resistance: Maximum 343 Pa
- KN95 Inhalation Resistance: Maximum 350 Pa
While this difference is small, healthcare workers wearing respirators for extended shifts may notice the variation in breathability, particularly during physically demanding activities.

CDC Guidance on Respirator Selection
CDC Protection Hierarchy
The CDC has published clear guidance on respiratory protection hierarchy, providing a framework for understanding relative protection levels across different mask types.
From Lowest to Highest Protection:
Lowest: Loosely Woven Cloth Products
Provide minimal filtration and poor facial seal. Not recommended for healthcare settings.
Low-Moderate: Layered Finely Woven Products
Multi-layer cloth masks offer improved filtration over single-layer options but still lack proper seal.
Moderate-High: Well-Fitting Disposable Surgical Masks and KN95s
Surgical masks provide better filtration, while KN95 respirators offer even more protection with improved facial seal.
Highest: Well-Fitting NIOSH-Approved Respirators (Including N95s)
Offer the highest level of protection with certified filtration and proper facial seal when fit-tested.
CDC Recommendations for KN95 Use
While N95 respirators provide the highest level of protection, CDC officials have recommended upgrading to KN95 masks as a significant improvement over surgical or cloth masks, particularly during periods of high community transmission.
When KN95 Respirators Are Appropriate
The CDC recognizes KN95 respirators as acceptable alternatives to N95s in many situations, particularly when:
- NIOSH-approved N95 respirators are in short supply
- Enhanced protection beyond surgical masks is needed
- Proper fit testing and seal checks can be implemented
- Healthcare workers need reliable respiratory protection during community outbreaks

Safety and Reliability
When properly fitted and worn correctly, KN95 masks provide reliable respiratory protection that significantly exceeds the capabilities of surgical or cloth masks. The CDC classifies them as respirators, meaning they must meet rigorous filtration standards.
Real-World Validation
The credibility of KN95 respirators was demonstrated when the U.S. House of Representatives transitioned from surgical masks to KN95 respirators for congressional staff following COVID-19 outbreaks in January. This institutional decision reflected confidence in KN95 respiratory protection for high-risk environments.
3M's Equivalency Statement
According to 3M, a leading respirator manufacturer with extensive expertise in respiratory protection, it is "reasonable to consider China KN95 as 'equivalent' to US NIOSH N95 and European FFP2 respirators" in terms of filtration performance.
This equivalency statement from a trusted manufacturer helps procurement managers understand that KN95 respirators, when sourced from reputable suppliers, offer comparable filtration performance to N95 respirators.
Key Performance Similarities
95% Filtration
Both N95 and KN95 respirators filter at least 95% of airborne particles at 0.3 microns, the most penetrating particle size.
Facial Seal Design
Both types are designed to create close facial seal, unlike surgical masks which have gaps around edges.
Respiratory Protection
Both protect the wearer from inhaling airborne hazards, not just preventing outward particle spread.
Electrostatic Filtration
Both use electrostatic filter media to capture particles efficiently without excessive breathing resistance.
Procurement Best Practices
The CDC provides guidance on identifying counterfeit respirators—a critical concern given the proliferation of fraudulent products in the marketplace. When purchasing KN95 masks for your healthcare facility or organization, implementing rigorous quality verification protects your staff and patients.
Essential Procurement Steps
Follow these best practices when evaluating KN95 respirator suppliers:
- Verify Published Filtration Efficiency Data: Request independent third-party test reports documenting ≥95% filtration efficiency at 0.3 microns
- Implement Fit Testing Protocols: Establish fit testing programs for all users to ensure proper seal regardless of respirator type
- Request Product Samples: Never commit to large orders without testing samples first. Evaluate comfort, breathability, and seal quality
- Evaluate Headband Design: Quality construction includes durable elastic or cloth headbands that maintain tension over extended use
- Obtain Third-Party Test Reports: Reputable manufacturers provide independent laboratory filtration testing from recognized testing facilities
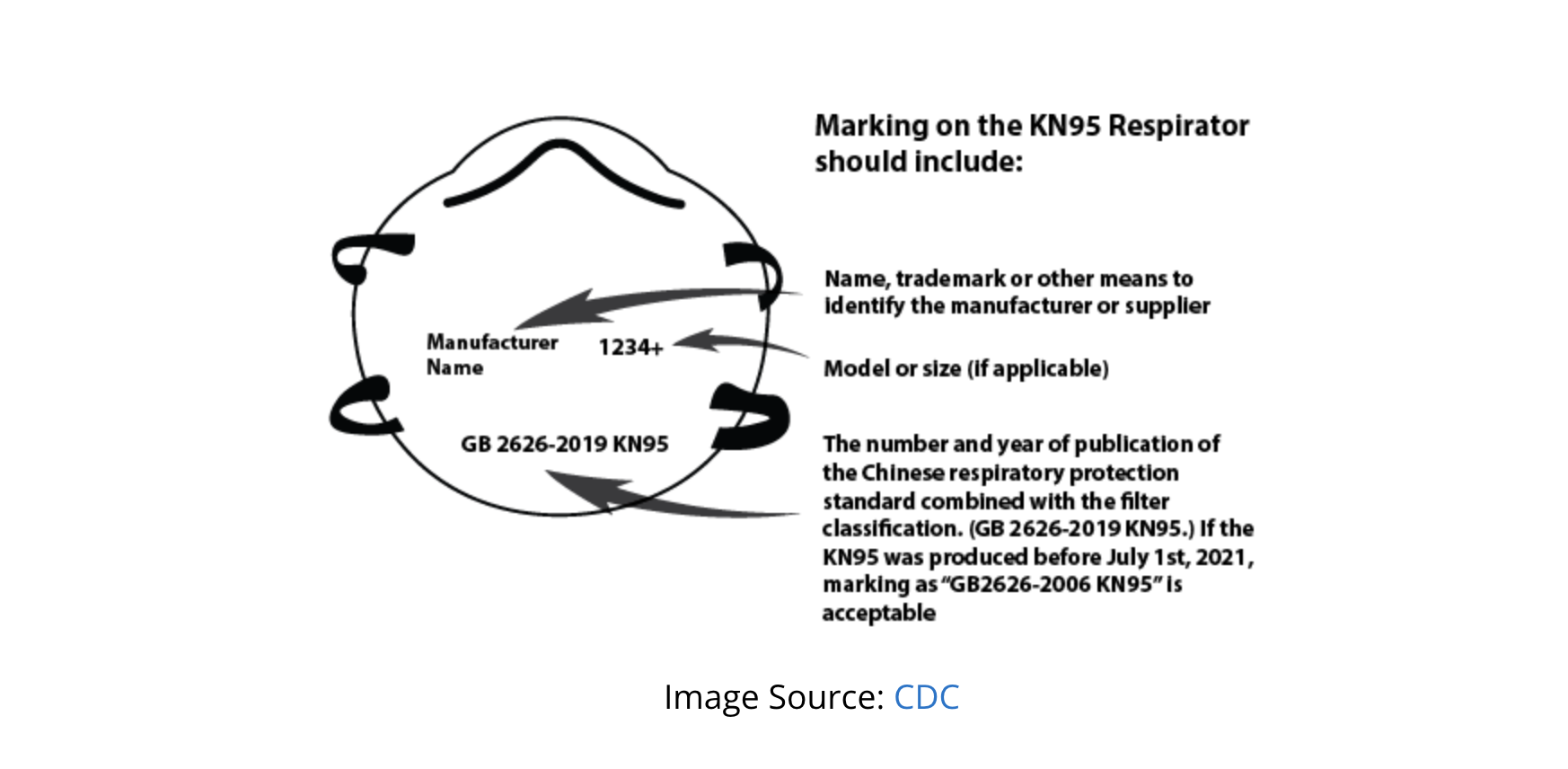
- Confirm Proper Markings: Verify that KN95 designation, manufacturer information, and relevant standards appear on both the product and packaging
- Assess Supplier Credibility: Work with established distributors who can document supply chain and provide manufacturer verification
Red Flags to Watch For
Warning Signs of Counterfeit or Substandard Products
- Prices significantly below market rates for comparable products
- Suppliers unable or unwilling to provide filtration test reports
- Missing or incorrect markings on respirators or packaging
- Vague or inconsistent information about manufacturing source
- No clear chain of custody from manufacturer to distributor
- Resistance to providing samples before large order commitments
- Claims that seem too good to be true (e.g., "FDA approved KN95"—the FDA does not approve KN95 respirators)
Making the Right Choice for Your Facility
Both N95 and KN95 respirators serve important roles in respiratory protection programs. Your selection depends on several factors specific to your organization's needs and circumstances.
Choose N95 Respirators When:
- Your facility requires NIOSH-approved respiratory protection for regulatory compliance
- You need the highest available standard of respiratory protection
- Supply availability and pricing are comparable to KN95 options
- Your organization has established N95 fit testing protocols and sized inventory
- OSHA or institutional requirements specifically mandate NIOSH-approved respirators
Choose KN95 Respirators When:
- N95 respirators are unavailable or in limited supply
- You need reliable respiratory protection that exceeds surgical mask performance
- Cost considerations are significant and KN95 offers better value
- You can verify product quality through reputable suppliers and testing
- Your application doesn't specifically require NIOSH-certified products
- You can implement proper fit testing regardless of respirator type
Your Trusted Source for Quality Respirators
Surgo Surgical Supply's Commitment to Quality
Surgo Surgical Supply provides certified KN95 respirators at competitive prices. We've partnered with verified suppliers to deliver high-quality respiratory protection, allowing you to focus on what matters most: patient care.
Our team understands the critical importance of respiratory protection in healthcare settings and the challenges Canadian facilities face in sourcing reliable PPE. We're committed to transparency, quality verification, and providing the information you need to make confident procurement decisions.
What Sets Surgo Apart
Quality Verification
We provide third-party filtration test reports and documentation for our KN95 respirators, ensuring you receive products that meet performance standards.
Competitive Pricing
Our established supplier relationships enable us to offer quality respiratory protection at prices that fit healthcare budgets.
Canadian Service
As a Canadian supplier, we understand the unique needs and regulatory environment of Canadian healthcare facilities.
Expert Guidance
Our knowledgeable team can help you select appropriate respiratory protection and implement effective PPE programs.
Ready to Source Quality Respiratory Protection?
Whether you need N95 or KN95 respirators, Surgo Surgical Supply provides the quality, documentation, and service Canadian healthcare facilities require.
Contact Surgo Surgical Supply today to discuss your respiratory protection needs, request product samples, or place an order. Our team is here to help you protect your staff and patients.

