
Not all biological indicators are interchangeable. The sterilizer type, cycle parameters, and the time available before results are needed all influence which BI is appropriate for a given application. This guide walks through the Solventum Attest™ steam BI product line to help sterile processing professionals identify the right indicator for their specific setup.
What Is a Biological Indicator?
A biological indicator (BI) is a testing system containing a defined population of highly resistant bacterial spores. For steam sterilization, BIs contain spores of Geobacillus stearothermophilus, the most resistant organism to moist heat. The logic is straightforward: if a sterilization cycle is sufficient to destroy these resistant spores, it is sufficient to destroy the less-resistant microorganisms present on clinical instruments.
A biological indicator is the only test that directly measures the lethality of a sterilization cycle. Chemical indicators and physical parameters confirm that conditions were present, only a BI confirms that those conditions were sufficient to kill resistant organisms.
The Three Tiers of Attest™ Steam BIs
Super Rapid Readout (24 Minutes) — Recommended for Most Facilities
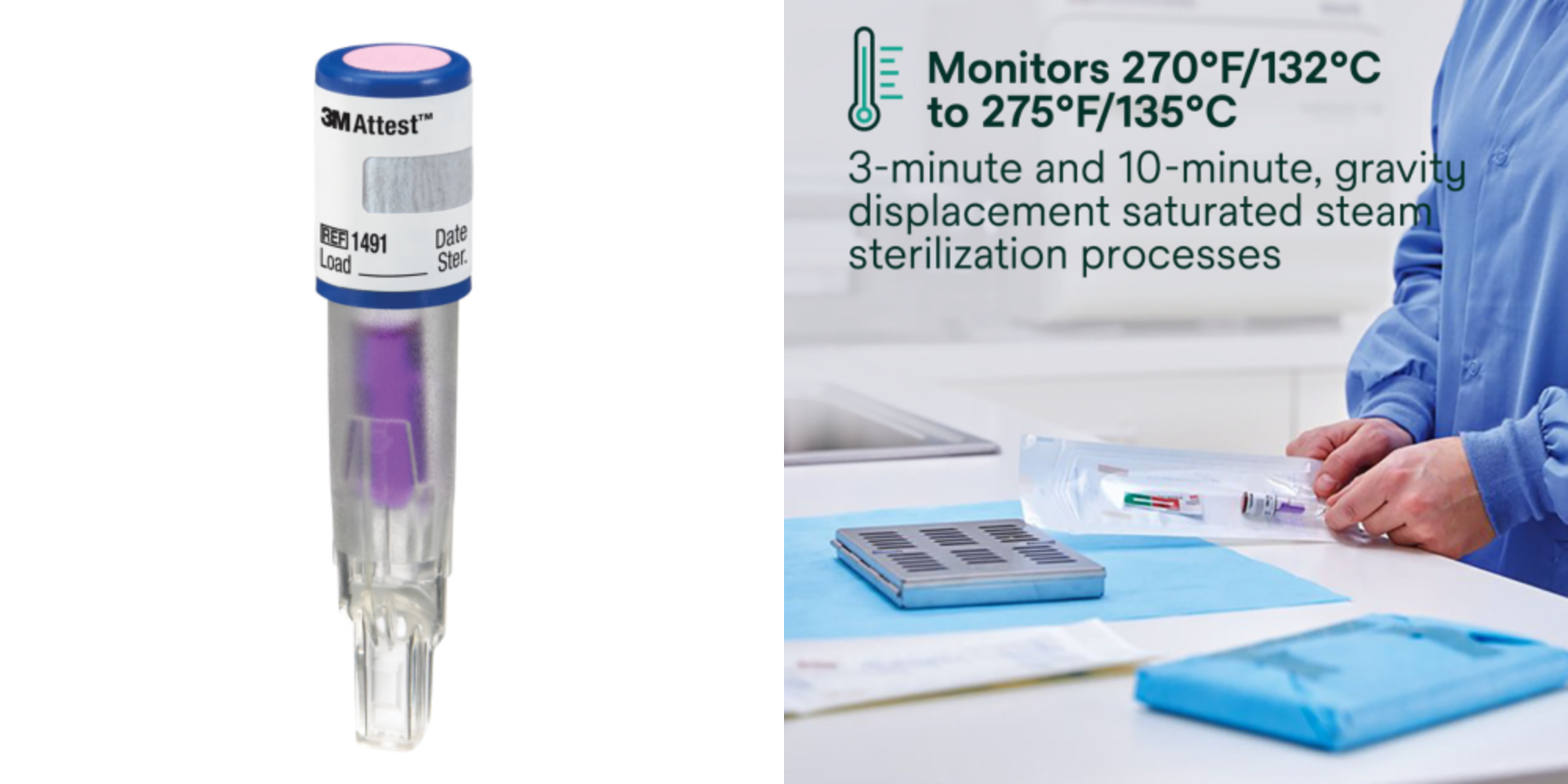
The Attest™ Super Rapid Readout Biological Indicators (Cat. 1491 for gravity cycles; Cat. 1492V for dynamic air-removal/pre-vacuum and SFPP cycles) return a fluorescent result in 24 minutes when used with the Attest™ Auto-reader 490M or 490/490H (with software version 4.0.0 or greater).
This speed enables Every Load Monitoring, a practice recommended by sterilization standards — without disrupting clinical workflow. The 1492V is validated for a wide range of pre-vacuum cycle parameters, including 270°F (132°C) at 3, 3.5, 4, 5.5, or 6 minutes, and 273°F (134°C) at 3 or 4 minutes.
Best for: Facilities seeking rapid confirmation, Every Load Monitoring, implant load monitoring, and clinics using the 490M auto-reader.
Rapid Readout (1 to 3 Hours)
The Attest™ Rapid Readout Biological Indicators (Cat. 1291, 1292, 1296) are read at 1 or 3 hours using the Attest™ Auto-reader 390. They cover gravity and vacuum-assisted cycles and are suitable for facilities where same-day results (rather than same-case results) are sufficient for their monitoring protocol.
Best for: Facilities with lower throughput or where next-case confirmation is not required, using the 390 auto-reader.
Standard (24 to 48 Hours)
Standard BIs (Cat. 1261, 1262, 1262P) are incubated using the Attest™ Biological Indicator Incubator 116 and require 24 to 48 hours for a result. These are appropriate for facilities with low sterilizer utilization or as a secondary validation tool.
Best for: Low-volume facilities, backup monitoring, or regulatory documentation purposes.
Matching BI to Cycle Type
The most important variable in BI selection is the sterilizer cycle type. Dynamic air-removal (pre-vacuum) sterilizers and gravity displacement sterilizers require different indicators. Always confirm that the BI you select is validated for your specific sterilizer's cycle temperature and time.
- Gravity cycles (e.g., 270°F/132°C, 3 or 10 min): Use Attest™ 1491 (Super Rapid), 1291 (Rapid), or 1261 (Standard)
- Dynamic air-removal / pre-vacuum cycles (e.g., 270°F/132°C or 273°F/134°C): Use Attest™ 1492V (Super Rapid), 1292 (Rapid), or 1262 (Standard)
- Low-temperature / 121°C gravity cycles: Use Attest™ 1493, 1493P, or 1262
Process Challenge Devices (PCDs)
CSA Z314:23 requires that BIs used for load monitoring be placed within a Process Challenge Device (PCD), a standardized challenge to the sterilization process that simulates a difficult-to-sterilize load. Solventum offers pre-assembled BI PCDs (e.g., Cat. 1492PCD-24, 1492PCD-05, 1493PCDG-24) that combine the BI and PCD in a ready-to-use format, simplifying daily monitoring procedures and reducing assembly time.
Need Help Selecting the Right Product?
Surgo's team can help you match the right Attest™ biological indicator to your sterilizer model, cycle parameters, and monitoring goals. Contact us or download the Solventum Attest™ Steam BI Product Selection Guide for a complete overview of catalogue numbers, indications for use, and compatible auto-readers.
Frequently Asked Questions
Q: What is a biological indicator and how does it work?
A: A biological indicator (BI) is a testing system containing a defined population of highly resistant bacterial spores — specifically Geobacillus stearothermophilus for steam sterilization. After a sterilization cycle, the BI is incubated to determine whether those spores survived. If they did not, the cycle is considered lethal to the less-resistant microorganisms present on clinical instruments. Unlike chemical indicators or physical parameters, a BI is the only test that directly measures whether a sterilization cycle was sufficient to kill resistant organisms — not just whether sterilizing conditions were present.
Q: What is the difference between Super Rapid, Rapid, and Standard Attest™ biological indicators?
A: The three tiers differ primarily in time to result and the auto-reader required. Super Rapid Readout BIs (Cat. 1491, 1492V) return a fluorescent result in 24 minutes when used with the Attest™ 490M or 490/490H auto-reader, enabling Every Load Monitoring without disrupting clinical workflow. Rapid Readout BIs (Cat. 1291, 1292, 1296) return results in 1 to 3 hours using the Attest™ 390 auto-reader, suitable for facilities where same-day confirmation is sufficient. Standard BIs (Cat. 1261, 1262, 1262P) require 24 to 48 hours using the Attest™ Incubator 116, and are appropriate for low-volume facilities or secondary validation purposes.
Q: How do I know which biological indicator is right for my sterilizer?
A: The most important factor is your sterilizer's cycle type. Dynamic air-removal (pre-vacuum) sterilizers and gravity displacement sterilizers require different BIs — they are not interchangeable. For gravity cycles at 270°F/132°C, use Attest™ 1491, 1291, or 1261. For dynamic air-removal/pre-vacuum cycles at 270°F/132°C or 273°F/134°C, use Attest™ 1492V, 1292, or 1262. For low-temperature gravity cycles at 121°C, use Attest™ 1493, 1493P, or 1262. Always confirm the BI you select is validated for your specific sterilizer's cycle temperature and exposure time before use.
Q: What is a Process Challenge Device (PCD) and is it required?
A: A Process Challenge Device (PCD) is a standardized device designed to simulate a difficult-to-sterilize load, creating a defined challenge to the sterilization process. CSA Z314:23 requires that biological indicators used for load monitoring be placed within a PCD, not run loose in the chamber. Solventum offers pre-assembled BI PCDs, including Cat. 1492PCD-24, 1492PCD-05, and 1493PCDG-24, which combine the BI and PCD in a ready-to-use format to simplify daily monitoring and reduce manual assembly time.
Q: Can the Attest™ 1491 and 1492V biological indicators be used with auto-readers other than the 490M?
A: Yes. The Attest™ 1491 and 1492V Super Rapid Readout Biological Indicators are compatible with the Attest™ Mini Auto-reader 490M as well as the standard Attest™ Auto-reader 490 and 490H, provided the 490/490H is running software version 4.0.0 or greater. All three readers deliver a 24-minute fluorescent result with these indicators. Facilities upgrading to the 490M from an existing 490/490H system do not need to change their BI products.
Q: Which Attest™ biological indicator is best suited for Every Load Monitoring?
A: The Attest™ Super Rapid Readout Biological Indicators, Cat. 1491 for gravity cycles and Cat. 1492V for dynamic air-removal/pre-vacuum and SFPP cycles — are the recommended choice for Every Load Monitoring. Their 24-minute result time, when used with the Attest™ 490M or 490/490H auto-reader, makes it practical to confirm every sterilization cycle before instruments are needed clinically. They are also the required indicator type for monitoring loads containing implantable devices, where CSA Z314:23 mandates quarantine until BI confirmation.
3M and Attest are trademarks of 3M. Solventum, the S logo, and other trademarks are trademarks of Solventum or its affiliates. BSI Kitemark is a trademark of BSI. Content prepared for surgo.com. © Solventum 2026.



.png)





