Why 24-Minute Biological Indicator Results Are a Game-Changer for Sterile Processing

For sterile processing professionals, few decisions carry more weight than when to release a sterilized load for clinical use. Release too early, before biological indicator results are confirmed, and you accept risk. Wait too long, and you create bottlenecks that delay surgical schedules and frustrate OR staff. For years, this tension was built into the job. Today, it doesn’t have to be.
The Problem with Traditional Biological Indicators
Standard biological indicators (BIs) used with conventional incubators require 24 to 48 hours before a result is available. In practice, this means most facilities release loads based on physical parameters and chemical indicator pass results alone, reserving BIs for documentation and after-the-fact confirmation.
That approach is compliant with minimum standards, but it doesn’t represent best practice. Every Load Monitoring (ELM), in which a biological indicator PCD is run and confirmed before every load is released, is the gold standard. Yet ELM adoption has historically been constrained by one thing: the time it takes to get a result.
How Rapid Readout Technology Changes the Equation
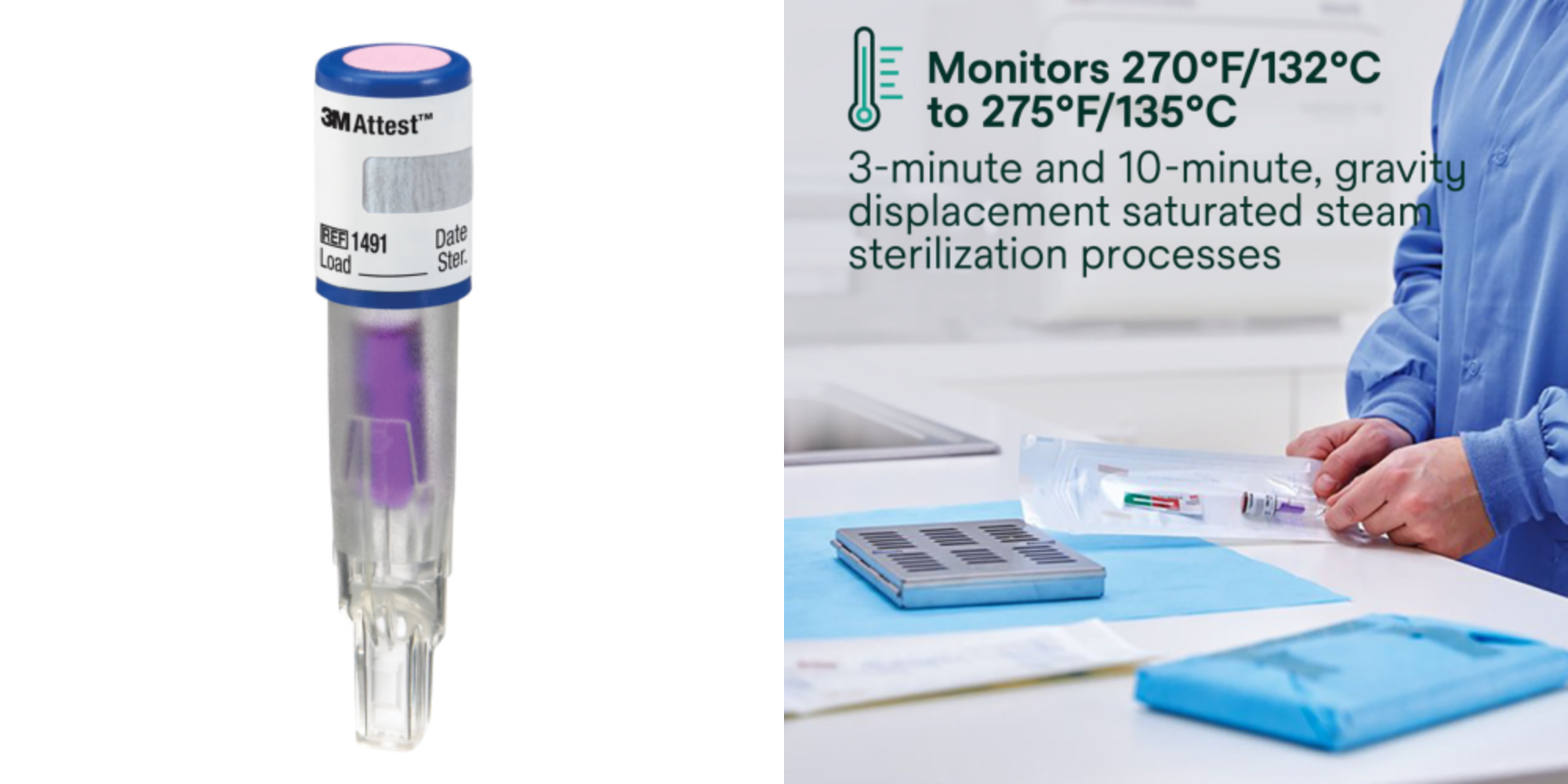
The 3M™ Attest™ Super Rapid Readout Biological Indicators, when used with the Attest™ Mini Auto-reader 490M, return a fluorescent result in 24 minutes. That’s fast enough to confirm a result before instruments are needed in the next case, particularly in facilities with normal turnaround times between procedures.
The 490M incubates at 60°C and uses fluorescent detection to identify whether spores of Geobacillus stearothermophilus, the highly resistant organism used in steam BI testing, survived the sterilization cycle. A positive result (growth) triggers a clear alert. A negative result (no growth) confirms cycle efficacy.
What 24-Minute Results Enable
- Every Load Monitoring without workflow disruption — BI results are available before the load is needed clinically in most routine scheduling scenarios.
- Faster quarantine release for loads containing implantable devices, where CSA Z314:23 requires BI confirmation before release.
- Reduced reliance on retrospective monitoring, where results arrive after instruments are already in use.
- Stronger documentation and traceability, especially when paired with Attest™ Connect Software for electronic record-keeping.
Practical Considerations for Your Facility
Transitioning to rapid readout BI monitoring requires minimal process change. The 490M is small, approximately five times smaller than the standard Attest™ 490, and operates via a simple plug-in workflow. Staff place the activated BI into a well, and the reader handles incubation and readout automatically, displaying a countdown on its large LED display.
For facilities already using Attest™ biological indicators, upgrading to the 490M typically requires no change to BI products, the 1491 and 1492V are compatible with both the 490M and the standard 490/490H auto-readers.
The Bottom Line
24-minute BI results remove the primary barrier to Every Load Monitoring. For sterile processing departments committed to the highest standard of patient safety, the 3M™ Attest™ Mini Auto-reader 490M makes ELM practical, not aspirational.
Frequently Asked questions
Q: What is Every Load Monitoring (ELM) and why does it matter?
A: Every Load Monitoring (ELM) is the practice of running a biological indicator Process Challenge Device (PCD) with every sterilization cycle and confirming the result before releasing the load for clinical use. It is broadly recognized as the gold standard for sterilization quality assurance because it provides direct, cycle-by-cycle confirmation of sterilizer efficacy — rather than relying on periodic spot-checks. ELM is particularly important for loads containing implantable devices, where CSA Z314:23 requires BI confirmation before release.
Q: Why do most facilities not practice Every Load Monitoring with standard biological indicators?
A: Standard biological indicators used with conventional incubators take 24 to 48 hours to return a result. In most clinical settings, instruments are needed long before that window closes, so facilities release loads based on physical parameters and chemical indicator results alone, using BIs for retrospective documentation rather than real-time release decisions. The time barrier, not compliance intent, has historically been the primary obstacle to ELM adoption.
Q: How does the 3M™ Attest™ Mini Auto-reader 490M enable Every Load Monitoring?
A: The Attest™ Mini Auto-reader 490M incubates and reads Attest™ Super Rapid Readout Biological Indicators at 60°C using fluorescent detection, delivering a confirmed pass or fail result in 24 minutes. That is fast enough to confirm a result before instruments are needed in the next case under normal surgical scheduling, removing the primary workflow barrier to ELM. Facilities no longer need to choose between releasing loads without BI confirmation and holding up the OR.
Q: What organism is used in steam biological indicator testing and why?
A: Steam biological indicators contain spores of Geobacillus stearothermophilus, chosen specifically because of its exceptional resistance to moist heat. If a sterilization cycle is sufficient to destroy these highly resistant spores, it is considered sufficient to destroy the less-resistant microorganisms present on clinical instruments. This makes the BI the only test that directly measures the lethality of a sterilization cycle, rather than simply confirming that sterilizing conditions were present.
Q: Do facilities need to change their biological indicator products to use the 490M?
A: In most cases, no. The Attest™ Mini Auto-reader 490M is compatible with the Attest™ Super Rapid Readout Biological Indicators 1491 (gravity cycles) and 1492V (dynamic air-removal/pre-vacuum cycles) — the same indicators used with the standard Attest™ 490 and 490H auto-readers. Facilities currently using these BIs can upgrade to the 490M without changing their BI procurement or retraining staff on new indicator products.
Q: What are the CSA Z314:23 requirements for biological indicator monitoring of implant loads?
A: CSA Z314:23 requires that every load containing implantable devices be monitored using a biological indicator Process Challenge Device, and that the load be quarantined — held from clinical use — until the BI result is confirmed negative. This is a mandatory requirement of the standard, not a best practice recommendation. With standard BIs taking 24 to 48 hours, facilities face regular pressure to release implant loads before confirmation. The 490M's 24-minute result time brings the quarantine window into alignment with realistic pre-procedure timelines.


.png)


